Neurobiology 301:
|
After the initial pride and sense of validation that comes with being accepted to UW's highly selective neurobiology major, I discovered in the first quarter of coursework that it is selective for a reason. The lab component of NBIO 301 was definitely my most challenging coursework of that quarter, and possibly of my entire undergraduate career (with only the possibility that it might have been surpassed by the next great challenge - NBIO 302). Over a mere ten weeks, I went from never having prepared my own experiments before to dissecting an entire crayfish and trying desperately to insert a micro-electrode into a single neuron to record the electrical activity. We had to write four full lab reports in standard scientific journal format with original data and figures, and in the final week we gave oral presentations also based on original data and figures. If you're wondering how difficult this is, consider that many PhD candidates spend years gathering and analyzing data for a single scientific paper. Through many painful lab sessions struggling to obtain data, sleepless nights navigating our LabChart program and fighting to make clear figures in Microsoft word, as well as days wondering if I was really cut out for this, I learned how to improvise and work with the unexpected (which I would later discover to be a very important research skill outside of the classroom), how to get over the fear of making mistakes during dissections, and how to present my data and analyze it critically. I also learned how much inclusion/exclusion selectivity and interpretation goes into the supposedly "scientific" process of research, which has made me a more critical reader of papers published in scientific journals. Below you can see the figures (the first document) and lab report (the second document) I wrote for our fourth lab - focusing on cellular and molecular neurobiology in the crayfish.
Excerpt:
The results from Experiment 6 showed that the recovery time of EPSP amplitude was dependent on the frequency of the applied current pulses. This is important because it implies that higher frequency stimuli induce a longer-lasting change in the how the post-synaptic cell responds to isolated (low frequency) stimuli. This means that high frequency stimuli induce longer periods of facilitation, which may be necessary for memory integration and learning.
The results from Experiment 6 showed that the recovery time of EPSP amplitude was dependent on the frequency of the applied current pulses. This is important because it implies that higher frequency stimuli induce a longer-lasting change in the how the post-synaptic cell responds to isolated (low frequency) stimuli. This means that high frequency stimuli induce longer periods of facilitation, which may be necessary for memory integration and learning.
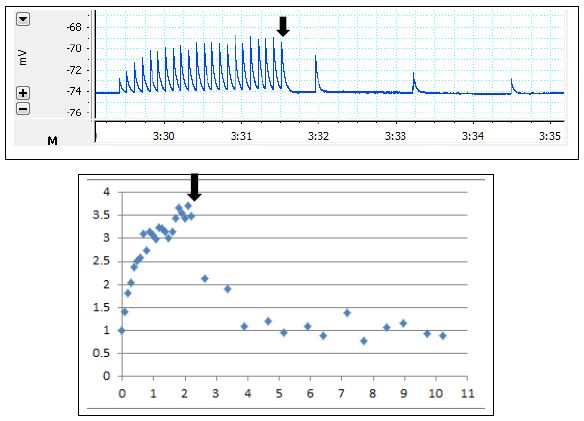
Figure 5.1: Time Course of Facilitation in Crayfish Neuromuscular Junction. Intracellular recording of post-synaptic potentials in a crayfish SFM cell. Voltage (in mV) is shown on the y-axis. Time (in seconds) is shown on the x-axis. In this preparation, the crayfish nerve had been crushed between the nerve cord and the extracellular suction electrode. We used a pulse stimulator to apply pulses of current at a frequency of 10Hz for periods of 2.2 seconds while simultaneously using a function generator to continuously apply pulses of current at a frequency of 0.787Hz. The resulting EPSPs that were recorded are shown in Box A. We then averaged data from 2 trials to produce a facilitation plot for this frequency of stimulation (Box B). The plot shows the facilitation index of each EPSP as a function of time. The downward arrow marks the time at which we stopped applying pulses of current from the pulse stimulator. Note that the pattern of the data changes at this point from increasing to decreasing facilitation index over time; the first EPSP peak was 1.367 mV.
|
Visit other pages on this pathway:
|
|